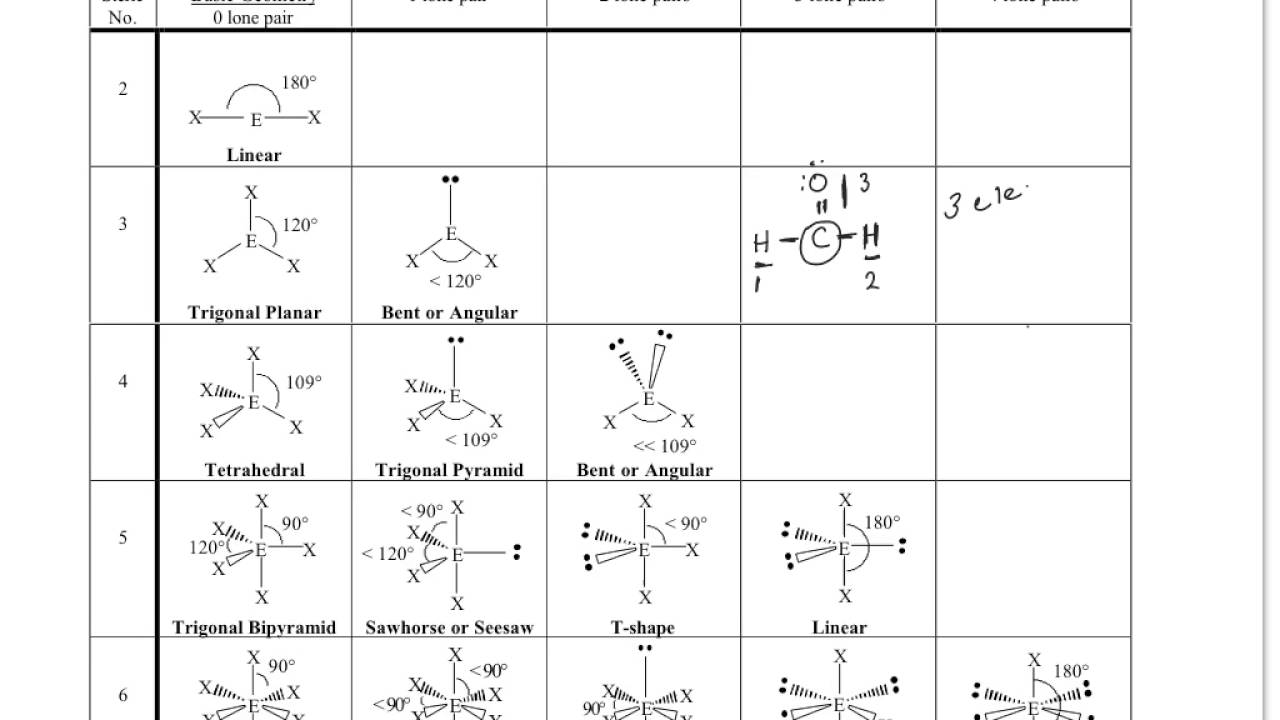 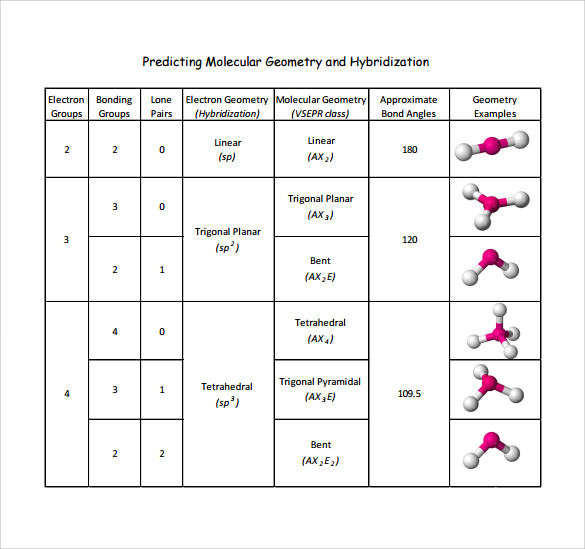 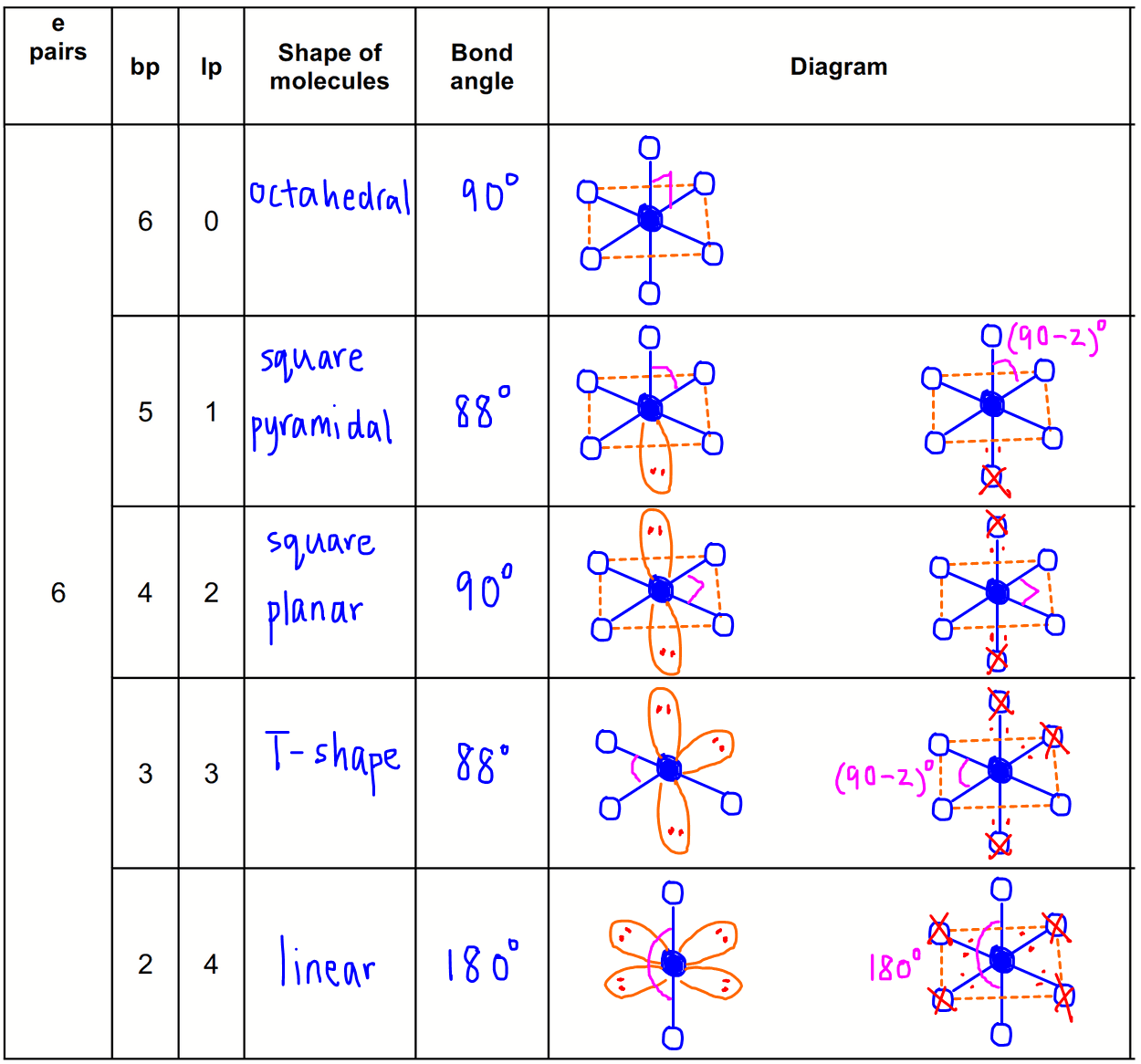
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. The chart below depicts some molecule geometries based on the number of electron pairs present. A solution to this problem was proposed by Linus Pauling, who argued that the valence orbitals on an atom could be combined to form hybrid atomic orbitals. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion It is difficult to explain the shapes of even the simplest molecules with atomic orbitals.In NH 3, for example, N, with a 2s 2 2p 3 valence electron configuration, can hybridize its 2s and 2p orbitals to produce four sp 3 hybrid orbitals. The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral. The bonding in molecules such as NH 3 or H 2 O, which have lone pairs on the central atom, can also be described in terms of hybrid atomic orbitals. The VSEPR chart confirms that a molecule with AX 3 generic formula possesses an identical electron and molecular geometry or shape, i.e., trigonal planar, as we already noted down for the benzene (C 6 H 6) molecule.Molecular geometries take into account the number of atoms and the number of lone pair electrons.It is the simplest aldehyde made up of two hydrogens, one carbon, and one oxygen. Formaldehyde is an organic compound that appears as a colorless gas with the chemical formula CH2O. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. Formaldehyde (CH2O) lewis dot structure, molecular geometry, polar or non-polar, hybridization. as per VSEPR theory, this carbon 1 holds the electron and molecular geometry of tetrahedral. It gives information about the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. Geometry of the water molecule with values for O-H bond length and for H-O-H bond angle between two bonds. Using VSEPR theory and its chart, we can easily determine the shape or geometry of CH3COOH. The O-C-N atoms lie in a planar arrangement, on a straight line therefore the O-C-N bond angle is 180° in OCN. Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. The OCN ion has an identical electron and molecular geometry or shape i.e., linear.The geometry of the regions of electron density is roughly the same as what we see when no NB pairs are involved. \): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. Molecules with 1 NB pairs and only single bonds.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
